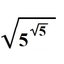Science topic
Molecular Dynamics Simulation - Science topic
Explore the latest questions and answers in Molecular Dynamics Simulation, and find Molecular Dynamics Simulation experts.
Questions related to Molecular Dynamics Simulation
Good morning, I am carrying out steam distillations with urea/water solutions on fresh rosemary, urea has a relatively low vapor pressure and in theory the vapors generated by this solution should be exclusively water. I got a yield per kg of 3ml EO which would normally be 0.7. Systems called deep eutectic solvents are likely to form between the essential oil/water/urea compounds. It was my intention to study the phenomenon with molecular dynamics, but I don't know which force field to use for the phase changes and the parameters (times, temperatures, etc.)
Curiosity: if you put a 50% water/urea solution in a beker, after a few days the urea recrystallizes coming out of the beker (see image)

Dear Amber user,
I want to study the interactions between protein and ions. I am running a simulation with protein, ions and water and I want to save the trajectories of protein and ions.
I know there is a ntwprt command that can allow Amber to save coordinates from atom number 1 to ntwprt, but however, in the topology, the order of atoms comes from protein, waters, and then ions. I can't use this ntwprt command to save protein and ions only.
I wonder if there is a way to save these two trajectories?
Thanks for any help.
I have two groups defined in the index file. The group [ protein ] has all protein atoms and the group [ DNA_5 ] has all the atoms of residue id 5 of DNA. I want to calculate the distance between center of mass of [ DNA_5 ] and each residue of [ protein ]. These distance will be given w.r.t time, i.e., say there are 5 protein residues, I should get 5 distances (each residue-DNA_5 distance) for each snapshot.
Is it possible to do by gmx distance directly? If not please suggest me a possible script !!
I am a beginner in molecular dynamics, it would be of great help if someone could help me in rectifying the problem.I am attaching the input files along with the dump file for reference.


I am running simulations with oxdna using advanced techniques of sampling as Umbrella Sampling.
while trying to generate CGenFF topology for Ligand after extracting from docked file, i got error which i'm showing as a picture. What could be the possible reason and how will i rectify it for smooth running of MD simulation.

Hi,
I've noticed a common practice in publications where multiple Molecular Dynamics (MD) simulations are conducted with varying initial velocities. However, I'm uncertain about the appropriate method for analyzing the resulting data. Could someone please provide guidance on this matter?
To clarify, let's say I have files named md1.xtc, md2.xtc, and md3.xtc. I understand that I can perform individual analyses such as Root Mean Square Deviation (RMSD) and Root Mean Square Fluctuation (RMSF) for each simulation and display them on the same graph. Alternatively, I could calculate the average RMSD values along with standard deviation and represent them on the graph.
Moving forward, I intend to conduct MD simulations involving ligands. My plan is to derive an average structure from these simulations and apply docking. My question is: does it make a difference which simulation I choose to continue with for further analysis?
Moreover, I'm curious about the expected presentation of these analyses when publishing an MD simulation study. Could anyone share their experiences and insights on these matters?
Thank you in advance for your assistance.
In a coarse grained model, such as DPD (dissipative particle dynamics) or MD(molecular dynamics), how to calculate the entropy and the interaction energy so to calculate the free energy?
Fatal error:
Atom HD1 in residue HIS 822 was not found in rtp entry HSE with 17 atoms while sorting atoms.
For a hydrogen, this can be a different protonation state, or it
might have had a different number in the PDB file and was rebuilt
(it might for instance have been H3, and we only expected H1 & H2).
Note that hydrogens might have been added to the entry for the N-terminus.
Remove this hydrogen or choose a different protonation state to solve it.
Option -ignh will ignore all hydrogens in the input.
I also Followed the suggestion to using -ignh in the code but it give me this error:
Fatal error:
Atom OXT in residue VAL 961 was not found in rtp entry VAL with 16 atoms
while sorting atoms.
Would anyone please help me? I can't concentrate on my studies unless I solve them.
I am using a constant potential version of VASP (cp-VASP) for a restricted molecular dynamics simulation to study the dissolution process of Cu from the Cu(111) surface. Towards the later stages of the MD simulation, I observed a significant increase in the Gibbs free energy, and hydrogen atoms began detaching from water. Why is this happening? My simulation has not yet reached the target number of steps. Here is my plot, MD trajectory,and my INCAR. (Sorry, the previous graph of the Gibbs free energy was incorrect. I have uploaded the new one here.)Thank you in advance for your assistance
SYSTEM
ISTART = 0
ENCUT = 400
ISPIN = 1
ISMEAR = 1
SIGMA = 0.2
ALGO = VeryFast
ISYM = 0
LREAL = Auto
LWAVE = .FALSE
LCHARG = .FALSE.
NSW = 2000
NELM = 25
POTIM = 0.5
IBRION = 0
EDIFF = 1E-4
# MD Parameters
MDALGO = 2
SMASS = 0
TEBEG = 300 ; TEEND = 300
# VASPsol Solvent Model - This corresponds to water
LSOL = .TRUE.
EB_K = 80
LAMBDA_D_K = 3.0
# CP-VASP Parameters
LCEP = .TRUE.
NESCHEME = 2
CAP_MAX = 2.0
NEADJUST = 5
TARGETMU = -3.6
FERMICONVERGE = 0.05
# slow growth
LBLUEOUT =.TRUE
INCREM = 0.0025

i have performed simulation in two steps and I wasn't able to merge the energy files. Provide help
Command line:
gmx_mpi -merge -f XXX.gro heme.gro -o complex.gro
Error in user input:
Invalid command-line options
In command-line option -merge
Invalid value: heme.gro
In command-line option -merge
Invalid value: 33.gro
In command-line option -merge
Option specified multiple times
For more information and tips for troubleshooting, please check the GROMACS
I merged heme and a ligand by manually editing gro and top instead of “-merge”, after which MD was performed. This result was unexpected for me because heme is not covalent binding to the protein, and it and the ligand are separated from the protein in less than 10ns of MD, but the ligand can only be injected once, so how do I make sure that the protein and heme are integrated as a whole, and MD of the ligand with the conjugated protein I constructed?
I am trying to understand the influence of polymer films sliding against each other. For the simulation, I saw many molecular dynamics simulations, like LAMMPS, GROMACS, etc. I am new to this field. Can someone who has experience with these methods suggest which one is the best one for the particular case?
Dear researcher,
I want to simulate my protein with the denaturants (MW-139 Da). I am using Gromacs 2018.2 version and the extra molecules were added by using gmx insert-molecule command.
I want to add different concentration of denaturants like 10, 20............. til 90%. I have successfully added the 20% denaturants in the simulation box and run the simulation while I want to add more than 20% so the command adds a particular number and terminate with a massage (2241 is successfully added out of 3271 requested).
I have increased the box size so the number of denaturants were also increased due to increase of water molecules, so problem is same. I also read the gromacs mailing list but i can not find any solution.
Please suggest me how i can add the total molecules in the simulation box.
Thanks for your response in advance.
In this figure, a pca plot is shown for different protein , protein complex systems depicting the essential subspace for these systems. I want to know how can I interpret these plots ? What information I can gain from it ?

- When I'm trying to use the antechamber in amber version 16, I couldn't proceed using it to generate parameters, topology and coordinate files as I got this type of error.
- The error mentioning that the acdoctoral mode is on : check and diagnosis the problem in input files.
- Can I get a proper guidance from someone to fix this problem or suggest me other ways or tutorial that can guide me to generate parameter topology and coordinate files in (prmtop/rst7) format for amber force field in order to prepare my non - standard small residues for molecular dynamic simulation.
thank you in advanced

That forces are the particular atoms/molecules forces due the effect of another atoms/molecules
I have a doubt for coarse grained md simulation. I have used martinize.py script to model the protein. But from here I do not understand on what basis it modelled the protein. There are 2 chain in the protein, So, it split chain A in 5 chains, 1: 3 atoms in 1 residue,2: 278 atoms in 35 residue, 3: 6 atoms in 2 residues, 3:278 atoms in 35 residues, 5: 3 atoms in 1 residues. and for chain B, 1060 atoms in 130 residues. Suppose I want to form residue wise beads with martini. Is it possible?
I had taken two receptors related to anti hypertensive activity and docked 25 ligands with them. Conceptual DFT,MD simulation, ADMET and trajectory analysis also done. Can It be published and in where?
i have performed MD simulation of a protein and I want to use TICA for dimensionality reduction, since it is better than PCA, using the dihedral angles of the protein. However, one parameter in TICA that must be selected is the lag time. How can i select this parameter? (i do not want to continue to do MSM)
Hi. I have a solution of polymer where the solvent is water. The polymer concentration is given in wt% unit. Can anyone tell me how I can calculate the number of water molecules in a simulation box where the box size is 6nm cubic?
I want to perform MD simulation of DMF using GROMACS by following the path of pdb2gmx command. But for that pdb file of DMF is required which is not provided in default gromacs directory.
Also, how can I generate its rtp file?
#moleculardynamicssimulation #freeenergylandscape#3dimensional
I have a research using MD simulation (using GROMACS) for proofing the binding stability of protein-protein interaction. To measure whether the interaction is stable, I wish to employ binding affinity analysis.
I am familiar with PRODIGY, a binding affinity predictor using network of contacts at the interface of a protein–protein complex to help calculate it (https://elifesciences.org/articles/07454). However, I realized that PRODIGY is usually being used for static binding affinity and not typically used in dynamical systems.
Regardless, I have successfully employed scripts that: (1) extracted PDB files from the trajectory file (xtc), and (2) calculate each PDB file's binding affinity using PRODIGY. I plotted the binding affinity vs time and got this as a result.
Is this a correct approach to binding affinity analysis? or should I employ different analysis?
I greatly appreciate your insight and comments regarding this topic. Thankyou.

I ran a molecular dynamics simulation for a system that consists of a peptide-protein complex, the binding happens at certain residues.
on calculating the MM-GBSA per residue energy decomposition using all the frames (1000 frame) I found that most of the residues participating in the binding have negative values.
only one residue in the peptide has a positive value however its binding residue has a negative value.
I double-checked the trajectory and the binding is stable during the course of the simulation.
is there an explanation for such a value, as I can not explain the reason behind it!
In a groundbreaking development shared within a Facebook group, the barriers between GROMACS and Windows have been successfully dismantled. The GROMACS community is elated to announce the triumphant porting of the renowned molecular dynamics simulation software to the Windows platform. This achievement not only broadens the user base but also opens up new possibilities for collaboration and exploration in the realms of computational chemistry and bioinformatics.
Traditionally confined to Unix-based systems like Ubuntu, GROMACS has been a staple in molecular dynamics simulations. However, a recent announcement within a Facebook group has unveiled a transformative milestone – the successful porting of GROMACS to the Windows platform. This breakthrough, which allows users to run GROMACS simulations on Windows via the command prompt, signifies a significant step towards increased accessibility and inclusivity.
Installation GROMACS on Windows:
The key to this newfound accessibility lies in the GROMACS installer tailored for Windows, available for download at the following link:
This installer simplifies the process, enabling users to seamlessly integrate GROMACS into their Windows environment and execute simulations directly through the command prompt. This is installed version of Gromacs 2020.3, both CPU and GPU version is available. In GPU version, you need to install latest NVIDIA CUDA driver and in case of windows 11, you need to put a dll file in the bin folder. https://www.dll4free.com/cufft64_10.dll.html
Technical Insight:
The successful porting of GROMACS to Windows raises questions about the technical intricacies involved in this process. While the Facebook announcement doesn't delve into the specifics, the installer provided indicates a streamlined approach to bring GROMACS functionality to Windows users. Researchers and enthusiasts keen on exploring the technical aspects of this achievement are encouraged to investigate the installer and potentially contribute to the ongoing development.
Performance Evaluation:
One of the critical aspects of this porting endeavor is the performance of GROMACS on the Windows platform. Researchers are encouraged to share their experiences and insights regarding the performance of GROMACS simulations on Windows, comparing it to the traditional Unix environment. Identifying any optimizations or challenges encountered during this process will contribute to the collective knowledge of the community.
Community Collaboration:
The Facebook group announcement not only celebrates the successful porting of GROMACS to Windows but also invites the community to actively engage in the ongoing discussions. Users are encouraged to share their experiences, feedback, and potential improvements related to running GROMACS on Windows. The provided link serves as a hub for collaboration, fostering a sense of community among researchers, developers, and enthusiasts interested in GROMACS on Windows.
Dear All,
Could you tell me how to run MD simulation by any software. I have DS, VMD in hand. But I try to run 30 nsec in DS that spent 6 days, is that right? The reviewer asked me to perform this procedure in 100 nsec, however, my computer can not complete this process. Could you let me know how set up all parameters and settings to finish them. Many thanks for your help with my deep heart.
I recently tried to use tools on charm gui website in preparation for molecular dynamics simulation and the website brings a prompt saying "No such ID" after openning up an account and getting a temporary password
I am new to Desmond simulations and I want to know how can I find the estimated time left for a simulation to be completed? my 2nd query is how to perform B-Factor analysis after performing simulation on Desmond? Any help in this regard will be highly appreciated.
Thanks
I need it uopn a reviewer's request to make a 500ns MD
So if I want to know what the structures are for different regions in the 2D projection plot of PCA analysis, how would I proceed? Any idea?
I am trying to calculate the mean squared displacement and have a C-code in Molecular Dynamics Simulation. I am having a hard time in getting the exact plot as proposed for a particular system. Can anybody have a look at it and suggest me where I am doing wrong in calculating the Mean Squared displacement as function of time.
I am trying to use VMD/NAMD to carry out a molecular dynamics simulation, and while running the simulation, task manager shows that only 30% of my GPU is being utilized. When VMD starts up, it only shows 4GB of VRAM detected, while my NVIDIA RTX 3080 has 10GB of VRAM. I installed the CUDA accelerated versions of VMD and NAMD, what do I do to fully use my GPU for MD simulations? My CPU is not bottlenecking the simulation, it has 12 cores/24 threads and is only at 5-10% utilization.
I have run a MD simulation for a protein-ligand system using AMBER. There are multiple binding poses for the ligand during the simulation. I want to find out some representative binding poses of the ligand. How can I achieve this? I have tried the dbscan clustering by calculating the pair RMSD of every snapshots and I think it is not a good way.
Dear Researchers,
I currently need to and must learn the Molecular dynamics simulation for polymer composites material (i.e. Interface analysis between matrix and reinforcement, rheological-structure-property relationship, thermodynamic-crystallization, etc.). Therefore, please give me any advice how to get fast and good understanding (including the best references related to MD simulation specific for polymer composites) because I start from zero for this MD method.
For the information, previously I have experiences in experimental research of polymer composite such as fiber reinforced polymer (Carbon fiber, glass fiber) for structural and EM shielding application.
Warm regards,
Asep Bustanil
Hello all,
I want to calculate interaction map for protein-ligand binding. This is to identify interactions, particularly the role of non-specific electrostatic interactions existing between the protein-ligand during the md simulation trajectory obtained using NAMD. Can anyone please guide me with steps to calculate it?
What is the nature of H-bonding in GROMACS ? Is it primarily electrostatic , or does it calculate the number of H-bonds based on the angle and distance criteria?
(I used the CHARMM36 force field for my MD simulation)
I am considering running an MD simulation of a protein with varying ligand concentrations. I think replica exchange molecular dynamics should be able to do that, but I have no idea how that is done.
Is it possible to get a tutorial or possibly other methods for running such a simulation?
Dear all,
I performed a simulation of a membrane-transporter and ligand for 50ns.
During the simulation, and as I expected, the ligand comes out of the transporter protein and does not come back. However, after the removal of PBC, the ligand jumps to the other side of the membrane which is not possible at all. With careful visualizing of the system using VMD, I am convinced it is a PBC problem in the mol stage. I put the commands I used for PBC removal and I also attached the movies from the clustering and mol stages.
I would appreciate any help.
gmx trjconv -s step7_production.tpr -n index.ndx -pbc whole -f small.xtc -o whole.xtc
gmx trjconv -s step7_production.tpr -n index.ndx -pbc nojump -f whole.xtc -o nojump.xtc
gmx trjconv -s step7_production.tpr -n index.ndx -pbc cluster -f nojump.xtc -o cluster.xtc
gmx trjconv -s step7_production.tpr -n index.ndx -pbc mol -f cluster.pdb -o mol.pdb -ur compact
Dear all,
I am currently working on the simulation study of a complex whose ligand cannot be automatically parameterized. I have been using AMBER to do the parameterization, and now I have obtained the .lib for the ligand. I want to load the parameter to the complex to prepare the input file for simulation. After changing the residue and chain name of the ligand in the complex PDB, I did:
tleap -f oldff/leaprc.ff99SB
>source leaprc.gaff
>loadamberparams deoxyFeb.frcmod
>loadoff deo.lib
>complex = loadpdb deoxyCom.pdb
and it added missing heavy atoms that are not included in the parameter lib. (the whole message is as tleap.txt)
how should I deal with the problem and prepare the input file for MD?
Hi experts,
I heard we can use Grand Canonical Monte Carlo (GCMC) or a combination of other ensembles and techniques like a third-party plugin or script by modifying the source code. Also, maybe the NVT ensemble can serve as a starting point for µVT simulation and then use a technique like semi-grand canonical Monte Carlo or Gibbs ensemble Monte Carlo, both of them require significant modifications to the simulation protocol and possibly the source code.
If there is a straightforward method, please tell me.
Best regards,
S.Ziaei
How can I change the number of amino acids in pdf files?
I need to change amino acid number for docking, molecular dynamics and... .
I use SWISSMODEL but it's not working good for me.
I have completed MD simulations of protein -DNA complex upto 200ns using Amber tools but when I am calculating its MM-PBSA I have the same error every time

I have designed 100 derivatives of indole and conducted molecular docking, MD simulation studies, ADMET analysis, and physicochemical analysis. Can this data be published as a research article? I would appreciate your suggestions on this matter.
How can I download DESMOND for molecular dynamics analysis from the website: https://www.deshawresearch.com/downloads/download_desmond.cgi/ ?
I have already tried filling out the form and so far I can't access the download link or receive any link by email. Has anyone had the same problem?
We know that Root Mean Square Deviation (RMSD) and Root Mean Square Fluctuation (RMSF) are both common metrics used in the analysis of molecular dynamics (MD) simulations, particularly in the context of proteins. So what is the exact difference between these two analysis ?
I'm trying to perform a triplicate of my molecular dynamics simulation. Some published works use this technique but do not specify which and where the change is made. If anyone can help me, I'd really appreciate it!
I have the model as attached. I want to have a simulation box containing water molecules to mimick fluid in the simulation. The other image shows TIP3P water addedd to the system. I am new in MD simulation. Any idea about this is appreciated. Thanks


Hello everyone!
I'm about to start a research grant on energy storage materials. The first task is to replicate the results of a paper where they used DFT and MD calculations (with VASP) to simulate the interaction of a gas adsorbing onto a Li slab. (Stephan L. Koch, Journal of Power Sources, 2015, DOI: 10.1016/j.jpowsour.2015.07.027, Pages 150-161)
I have little experience with DFT calculations (although with Gaussian), but none with MD and VASP. Additionally, my university doesn't have a license for VASP.
Could you suggest valid alternatives to VASP and provide some teaching materials on how to use the software for these types of calculations?
Thank you very much!
Hello,
I have performed a simulation of Sodium Montmorillonite swelling due to water adsorption. However, I am not sure if the simulation is correct. I followed the following steps:
1. A model of Na-MMT and water was created and both have energy minimized and geometry optimized.
2. In one case-1 the Na-MMT was cleaved in 001 surface and in case-2 the Na-MMT was not cleaved.
1. Adsorption of water molecules in the interlayer space using the adsorption locator module.
2. Energy minimization and geometry optimization using the Forcite module.
3. Then 200ps of dynamics simulation in NVT ensemble and the next 200ps on NPT ensemble using the Forcite module.
In case-1 the increase of interlayer space was visible due to water adsorption but when the simulation was performed with out water adsorption the the interlater space increased too.
In case-2 the interlayer space nor the basal spacing changed.
How to commence simulations in nanoscale? In which part of the code do we introduce the nano dimensions?
Hello Everyone!
I am working on a project of DNA regulation of Nanoenzymes and need to do simulations of nanoenymes. Can anyone tell me how to do MD simulations of inorganic nanoparticles (nanosomes) through GROMACS? I already read some literature about MD simulations of nanoparticles but specifically with GROMACS I couldn't find any literature about simulations.
Your answers will be much appreciated.
Thanks
How can we apply a constant force on a single atom for carrying out MD simulation using NAMD?
I want to calculate the mean square displacement of GaAs using CPMD. From the mean square displacement, I want to extract the configuration of atoms (e.g atomic position of Ga and As). Could anyone please give me some guidelines on how I can perform it? Should I use only the cp.x package or by other means? Your time and suggestions would be a great help for me.
Dear ResearchGate community,
I have recently conducted two separate 100 ns molecular dynamics simulations using GROMACS, each involving a small molecule. My analysis of hydrogen bonding patterns has revealed an unexpected observation: zero hydrogen bonding and an unusually large number of pairs within 0.35 nm. For instance, at a specific time point (e.g., time = 1.90108e-33 ns), the number of hydrogen bonds reported is >1110000000. I would like to know about such variations and how should I fix it, and if anyone has insights into the potential implications of this observation. I appreciate any feedback or experiences you can share on this matter.
; Include forcefield parameters
#include "./charmm36-mar2019.ff/forcefield.itp"
; Include ligand parameters
#include "crx.prm"
Thank you
I have installed M3GNet to do molecular dynamics,
but there aren't any manuals or guide to implement the program.
I am docking multiple ligands (new designed ligand) against my protein using Autodock Vina in Chimera. The results displayed in this program are somewhat strange, because in some cases the ligands with no hydrogen bond or fewer bonds has the most negative Score than the ligand with more hydrogen bonds!!? Please look at the picture to get my mean(The best model in ligand with 2 hydrogen bonds has a score -8.3, While the best model in ligand with one or zero hydrogen bond has score -8.7 and -10.1 respectively!).
I understand that checking with other software or tools like PyMOL or PDBSUM will better help to analyze the possible interactions, however since I have several ligands with almost similar score and interaction network or equal hydrogen bond numbers, I am curious to now how to pick the best one (based on the in silico analysis) among them. If any body has suggestion for this I will appreciated it.

Hello,
I am working on a protein that belongs to NUDIX family hydrolases and therefore requires Mg ion as a cofactor. But the protein that I have downloaded from PDB is an apoprotein. I want to include Mg ion also in my docking and MD simulations. I have schrodinger software. Can anyone suggest me how to proceed with this problem?
Dear all,
I would like to know if anyone has experience with doing MD simulation of nucleic acids or proteins in high density liquids.
I would appreciate if I could be guided to some reading material
The idea is to restrain the movement of Dna so that the effect of uv damage can be studied
Thank you
Ayesha
While trying to run MD simulation for 50ns, i got some errors read as
"WRITING EXTENDED SYSTEM TO RESTART FILE AT STEP 500000
WRITING COORDINATES TO DCD FILE output.dcd AT STEP 500000
WRITING COORDINATES TO RESTART FILE AT STEP 500000
FINISHED WRITING RESTART COORDINATES
WRITING VELOCITIES TO RESTART FILE AT STEP 500000
FINISHED WRITING RESTART VELOCITIES
REINITIALIZING VELOCITIES AT STEP 500000 TO 310 KELVIN.
TCL: Running for 25000000 steps
ETITLE: TS BOND ANGLE DIHED IMPRP ELECT VDW BOUNDARY MISC KINETIC TOTAL TEMP POTENTIAL TOTAL3 TEMPAVG PRESSURE GPRESSURE VOLUME PRESSAVG GPRESSAVG
ENERGY: 500000 1758.0188 1801.9004 2376.2868 32.0673 -57791.6942 4645.6399 0.0000 0.0000 12929.5296 -34248.2515 314.2067 -47177.7811 -34247.2801 314.2067 -547.9328 -2890.9435 156892.4304 -547.9328 -2890.9435
ERROR: Atom 4439 velocity is 4671.87 1153.77 2833.69 (limit is 5000, atom 327 of 328 on patch 18 pe 1)
ERROR: Atoms moving too fast; simulation has become unstable (1 atoms on patch 18 pe 1).
FATAL ERROR: Exiting prematurely; see error messages above.
FATAL ERROR: Exiting prematurely; see error messages above."
The error happens just after minimzation is complete and the running for 25000000 steps just started. Any recommendation to solve this problem?
Hi
Can you please explain to me about the need to do the PCA analysis in molecular dynamics simulation?
Thanks alot
I'm trying to perform MD simulation of protein-ligand complex. The step5_production.inp was set for 100ns. After running for around 30ns the program stop automatically. How will i modify the file so that the simulation restart from where it was stop. The last line from my output log file read as
"WRITING EXTENDED SYSTEM TO RESTART FILE AT STEP 13000000
WRITING COORDINATES TO DCD FILE step5_production.dcd AT STEP 13000000
WRITING COORDINATES TO RESTART FILE AT STEP 13000000
FINISHED WRITING RESTART COORDINATES
WRITING VELOCITIES TO RESTART FILE AT STEP 13000000
FINISHED WRITING RESTART VELOCITIES
LDB: ============= START OF LOAD BALANCING ============== 204045
LDB: ============== END OF LOAD BALANCING =============== 204045
Info: useSync: 0 useProxySync: 0
LDB: =============== DONE WITH MIGRATION ================ 204045
LDB: ============= START OF LOAD BALANCING ============== 204047
LDB: TIME 204047 LOAD: AVG 0.347167 MAX 0.364001 PROXIES: TOTAL 83 MAXPE 15 MAXPATCH 4 None MEM: 704.516 MB
LDB: TIME 204047 LOAD: AVG 0.347167 MAX 0.364001 PROXIES: TOTAL 83 MAXPE 15 MAXPATCH 4 RefineTorusLB MEM: 704.516 MB
LDB: TIME 204047 LOAD: AVG 0.347167 MAX 0.364001 PROXIES: TOTAL 83 MAXPE 15 MAXPATCH 4 RefineTorusLB MEM: 704.516 MB
LDB: ============== END OF LOAD BALANCING =============== 204047
Info: useSync: 0 useProxySync: 0
LDB: =============== DONE WITH MIGRATION ================ 204047"
Any recommendation will be highly helpful
I am currently trying to run a command prompt for MD simulation but came across this error:
C:\frohaisa\MD>namd2 runme.namd > simul.log
FATAL ERROR: tried before startup to read config file parameter that was not set
while executing
"---------input-----"
(file "runme.namd" line 1)

[INFO ] Running calculations on normal system...
[INFO ] Beginning GB calculations with /home/bio/anaconda3/envs/gmxMMPBSA/bin/sander
[INFO ] calculating complex contribution...
100%|##########| 101/101 [elapsed: 08:08 remaining: 00:00]
[INFO ] calculating receptor contribution...
100%|##########| 101/101 [elapsed: 08:16 remaining: 00:00]
[INFO ] calculating ligand contribution...
100%|##########| 101/101 [elapsed: 00:02 remaining: 00:00]
[INFO ] Beginning PB calculations with /home/bio/anaconda3/envs/gmxMMPBSA/bin/sander
[INFO ] calculating complex contribution...
0%| | 0/101 [elapsed: 00:00 remaining: ?][ERROR ] CalcError
/home/bio/anaconda3/envs/gmxMMPBSA/bin/sander failed with prmtop COM.prmtop!
If you are using sander and PB calculation, check the *.mdout files to get the sander error
Check the gmx_MMPBSA.log file to report the problem.
File "/home/bio/anaconda3/envs/gmxMMPBSA/bin/gmx_MMPBSA", line 8, in <module>
sys.exit(gmxmmpbsa())
File "/home/bio/anaconda3/envs/gmxMMPBSA/lib/python3.9/site-packages/GMXMMPBSA/app.py", line 101, in gmxmmpbsa
app.run_mmpbsa()
File "/home/bio/anaconda3/envs/gmxMMPBSA/lib/python3.9/site-packages/GMXMMPBSA/main.py", line 205, in run_mmpbsa
self.calc_list.run(rank, self.stdout)
File "/home/bio/anaconda3/envs/gmxMMPBSA/lib/python3.9/site-packages/GMXMMPBSA/calculation.py", line 142, in run
calc.run(rank, stdout=stdout, stderr=stderr)
File "/home/bio/anaconda3/envs/gmxMMPBSA/lib/python3.9/site-packages/GMXMMPBSA/calculation.py", line 625, in run
GMXMMPBSA_ERROR('%s failed with prmtop %s!\n\t' % (self.program, self.prmtop) +
File "/home/bio/anaconda3/envs/gmxMMPBSA/lib/python3.9/site-packages/GMXMMPBSA/exceptions.py", line 171, in __init__
raise exc('\n\n' + msg + '\n\nCheck the gmx_MMPBSA.log file to report the problem.')
CalcError:
/home/bio/anaconda3/envs/gmxMMPBSA/bin/sander failed with prmtop COM.prmtop!
If you are using sander and PB calculation, check the *.mdout files to get the sander error
Check the gmx_MMPBSA.log file to report the problem.
Error occurred on rank 6.
Exiting. All files have been retained.
Abort(1) on node 6 (rank 6 in comm 0): application called MPI_Abort(MPI_COMM_WORLD, 1) - process 6
Binding sites along with binding energy can be easily determined using ADT. However, MD simulations have been reported by researchers using Schrodinger Desmond. So I was wondering if there is a possibility of carrying out MD simulations using ADT and generating RMSD curves/plots.
I selected Md_0_10.gro file and then md_0_10.xtc file then I got something weird like this in the video in vmd, can someone pls suggest what's wrong in this step?
I would like to have GPU support for GROMACS, my GPU is Intel Iris Xe Graphics, so I was recommended to use SYCL support. So I would like to ask if anyone knows how to proceed to install SYCL-GROMACS complete building without errors? I'm using Ubuntu 22.04 by the way if it helps to clarify.
In order to reduce the influence between domains that can cause dimerization in MD simulations through biosensor structures, FL biosensors were isolated, structural prediction was performed with Alpha Fold 2, and MD was performed with Gromacs.
Now I want to put two PDB files together from the trajectory file, including the situation of a specific frame, and restore them back to a single intact protein. I divided the front and back based on a specific part of the linker domain. How can I put two proteins back together into a single PDB file?
It would be nice if I could use the built-in functionality of GUI tools such as pymol or the additional plug-in to proceed with the work.

I've finished processing phosphorylation on the protein model using the PyTMs plugin in PyMOL. I've tried various methods, but I get the fatal error below on pdb2gmx. Is there a solution?
---------------------------------------------------------------------
Program: gmx pdb2gmx, version 2023.2
Source file: src/gromacs/gmxpreprocess/pdb2gmx.cpp (line 1042)
Fatal error:
The residues in the chain MET1--ASP863 do not have a consistent type. The
first residue has type 'Protein', while residue TPO395 is of type 'Other'.
Either there is a mistake in your chain, or it includes nonstandard residue
names that have not yet been added to the residuetypes.dat file in the GROMACS
library directory. If there are other molecules such as ligands, they should
not have the same chain ID as the adjacent protein chain since it's a separate
molecule.
For more information and tips for troubleshooting, please check the GROMACS
website at http://www.gromacs.org/Documentation/Errors
Dear all,
I am currently working on a protein simulation with a heme group in the protein. I am wondering whether anyone can tell me how I can set it up in GROMCAS with charmm forcefile, since error shown when I generated topology files. Thank you.
When I perform Born-Oppenheimer Molecular Dynamics (BOMD) simulations of small organic clusters using the CP2K software in NVE condition, I notice that running multiple simulations from the same initial structure produces remarkably similar trajectories. I'm curious about the underlying reason for this behaviour and would like to know how to introduce velocity randomisation in CP2K. I also attached input file of BOMD simulation.
Is there any program to convert ssDNA sequences to possible three-dimensional conformation for MD simulations?
Edit: It is an aptamer generated against specific target
I want to analyze the biomedical properties of green synthesized nanoparticles.
Need for online tools to perform molecular dynamics simulation
I'm looking to calculate charges for my ligand and later run MD simulation on them. I ran into this error suddenly when it had worked in the past. I have Gaussian 16 installed.
My slurm.out
Net charge: -1
Net charge: -1
Charge and Multiplicity [0,1]? Percent or Route card? Percent or Route card? Percent or Route card? Percent or Route card? Route card? Titles? Titles? Charge and Multiplicity? ../../simulation_OTP111_3One_0_Emin.sh: line 25: /programs/g16: Is a directory
Error termination in NtrErr:
ntran open failure returned to fopen.
Error: segmentation violation
rax 0000000000000000, rbx 0000000005c10e60, rcx ffffffffffffffff
rdx 0000000000038f42, rsp 00007fffffa34778, rbp 00007fffffa347b0
rsi 000000000000000b, rdi 0000000000038f42, r8 00002aaaaaae8800
r9 00002aaaaaae8800, r10 00007fffffa33b60, r11 0000000000000206
r12 0000000000000000, r13 0000000005c04598, r14 00007fffffa39bd0
r15 00007fffffa39910
/lib64/libpthread.so.0(+0xf630) [0x2aaaab0ab630]
/lib64/libc.so.6(kill+0x7) [0x2aaaab5f0657]
/programs/g16/newzmat() [0x458fc5]
/programs/g16/newzmat() [0x49ccb9]
/programs/g16/newzmat() [0x4873f2]
/programs/g16/newzmat() [0x434284]
/programs/g16/newzmat() [0x411a36]
/programs/g16/newzmat() [0x4109a6]
/programs/g16/newzmat() [0x410864]
/lib64/libc.so.6(__libc_start_main+0xf5) [0x2aaaab5dc555]
/programs/g16/newzmat() [0x40afa9]
/scratch1/alex/glyco_project/glycomimetics/scripts/make_gaussian_input_file.sh: line 52: 233282 Aborted /programs/g16/newzmat -ichk -ozmat -prompt ${old_chk} ${gaussian_input} < newzmat_here_esp.txt
../../simulation_OTP111_3One_0_Emin.sh: line 31: /programs/g16: Is a directory
Num atoms : 19
Getting ESP charges on OTP111_3One_0_ligand_esp_g16.log
Running Resp
Error: No ESP fitting centers and fitting values exist, adding 'iop(6/33=2) iop(6/42=6)' to the key word list
/scratch1/alex/glyco_project/glycomimetics/scripts/Run.resp: line 42: /scratch2/alex/Set5/Trial_1/simulation/analog_OTP111_3One_0/1_leap/resp-2.2/resp: No such file or directory
Slurm script ends
It is possible to learn md simulation without any phd degree or advisor by learning form books and github tutorial and start own research in feild of material science and biochemisty and produce reaserch paper ?
If possibe then please provide me some ideas , advice and how can i start this journey.
currently i have basic knowlegde of software like lammps , namd ,openbabel and vmd ..+ some md simulation and qunatum chemisty knowledge
Hi everyone,
As you know, in MD simulation of a biological system, typically, one atom is moved, followed by recalculating forced, then another atom is moved and so forth. Do you know if any of the MD simulation tools, GROMACS, NAMD, AMBER, etc., allows us to run the simulation such that all atoms are moved a single step of the simulation before recalculating forces? There was a study that showed an more accurate results when doing so.
Thank you in advance!
--Ra'ed
How to generate ligand.itp and ligand.gro files for md simulation using amber14 force field?? Kindly help!!
Thanks in advance.
Investigating the potential for manipulating the movement of a <111> screw dislocation, typically prone to {110} slip, to instead traverse {112} slip planes by applying specific strains in Molecular Dynamics Analysis.
i have been doing the molecular dynamic simulation for 6lu7 protein and some ligands and constantly getting the namd2 error.
i submitted my article in where i conducted molecular dynamic simulation 100 ns for a complex protein-ligand. Reviewer asked us for more work to be done for this study as below
''More extensive simulations of longer simulations could make this article more appropriate. This could be done with structural visualization of clusters from non-parametric clustering or of structures from free-energy wells on PCA-based free-energy surfaces. And then significant discussion of the result protein structures and conformational changes".
can you please explain to me what he mean by non-parametric clustering and share with some articles examples that include example of what reviewer expect from us.
thannk you.
Hello everyone
I’m trying to run a MD simulation in acetonitrile as solvent but when I execute gmx-solvate command, solvent molecules break apart making the number of atoms in .gro and .top files different from one another. Does anyone know how this issue can be addressed? I appreciate any help in advance.
- I have downloaded acetonitrile solvent box from virtualchemistry.org
- The force field I’m using is OPLS.
I am running the MD simulations for 50000000 steps using GROMACS software but it has terminated (after 36000000 steps) before completion due to power off. I would like to continue the same calculation to complete the simulation.
Kindly explain how to calculate all the parameters listed in the image , the below data is for polyetehelyne.

Hello, I'm seeking clarification on the selection of suitable boundary conditions for simulating shear deformation of a screw dislocation using LAMMPS. In my script, I currently employ the following commands:
```lammps
fix 1 upper setforce 0.0 0.0 0.0
fix 2 lower setforce 0.0 0.0 0.0
fix 3 upper move NULL ${strainrate} NULL
fix 4 lower move NULL-${strainrate} NULL
fix 5 mobile nve
```
I have several uncertainties:
1. Should I fix all three degrees of freedom in fix 1 & 2 for shear deformation in the Y direction, or are specific degrees of freedom recommended?
2. In fix 3 & 4, should I use NULL or 0.0?
3. Should fix 5 be applied to only middle atoms or to all atoms?
Any insights would be greatly appreciated!

i want to perform md simulation of silica nanoparticles in water. can anyone help?any instruction or tutorial might help
I'm trying to generate ligand topology files through CHARMM FF but i'm getting this error. I installed the respective python version but error is still same
python cgenff_charmm2gmx.py UNK unk_fix.mol2 unk.str charmm36-jul2022.ff
You are using a Python version in the 2.x series. This script requires Python 3.0 or higher.
Please visit http://mackerell.umaryland.edu/charmm_ff.shtml#gromacs to get a script for Python 3.x
Kindly help
I want to perform a protein-ligand MD simulation using Amber. I have generated ligand frcmod, lib, rst7, and prmtop files. Then, I loaded my complex file, which contained the ligand and protein in water (generated using Packmol-memgen). However, when I tried to save the rst7 and prmtop files for the complex, I encountered this error: 'FATAL: Atom .R<***> does not have a type.' I've attempted to resolve this issue multiple times, but it keeps happening. I followed the tutorial exactly as outlined on the AmberMD website ('Simulating a pharmaceutical compound using Antechamber and the Generalized Amber Force Field'). Surprisingly, the problem persists. even though I tried to use the same molecule, protein, and files provided on the website to check out if there were any problem with my own files and again i ended up with same problem. Can anyone help me figure this out?"
Hello Everyone! I am doing protein-ligand MD simulations through MOE. But, can't read and understand their results neither I find any tutorial regarding this. Anyone please help me with this matter. Any tutorial about MD simulation through MOE?
Your suggestions are highly appreciated.
Thanks!


There are lots of simulation sofwares currently. Give the most one you use and explain the reasons so that others can take a reference if he or she wants to learn.
I'm working on molecular dynamic simulation of a protein-ligand complex. I tried using GROMACS, but it gives several errors whether I allow or ignore hydrogen eg "Atom HN1 in residue GLU 221 was not found in rtp entry GLU with 17 atoms
while sorting atoms.". NAMD and VMD seem complicated to me, and CHARMMS gives me errors
is there a way to refine my complex to prevent these errors? or is there a simpler MDS program/protocol I can use?
Dear all,
I have the cif file of the MOF structure, the atoms and their indexes goes like this:
C1 1.0 0.3521 0.68248 0.49724
Cr2 1.0 0.6727 0.73299 0.90674
C3 1.0 0.9812 0.86236 0.85294
C4 1.0 0.29174 0.5265 0.71863
...
C1185 1.0 0.17108 0.59341 0.61745
C1186 1.0 0.14289 0.58226 0.66194
C1187 1.0 0.1861 0.67126 0.70408
Cr1188 1.0 0.25915 0.66547 0.69122
...
But in the simulation, we divided the atoms to different types,e.g. only 3 types of carbon atom and 2 types of Cr and so on. The force field is made for the type of atoms such as:
C1 lennard-jones 47.8562 3.47299
C2 lennard-jones 47.8562 3.47299
Cr1 lennard-jones 7.54829 2.69319
Cr2 lennard-jones 7.54829 2.69319
So the data need to be like:
C1 1.0 0.3521 0.68248 0.49724
Cr2 1.0 0.6727 0.73299 0.90674
C1 1.0 0.9812 0.86236 0.85294
C2 1.0 0.29174 0.5265 0.71863
...
C1 1.0 0.17108 0.59341 0.61745
C1 1.0 0.14289 0.58226 0.66194
C2 1.0 0.1861 0.67126 0.70408
Cr2 1.0 0.25915 0.66547 0.69122
...
How do I change the atom index? There are too many atoms for me to do it manually. Is there any software that can do this ?
Dear all
I hope this message finds you well. I am currently working on molecular dynamics simulations using LAMMPS and have encountered an issue related to setting up bond interactions. I have consulted the paper and found the appropriate bond_style (harmonic) and the required parameters for each bond. However, I am facing difficulties in determining the correct order (as illustrated in the diagram below, labeled as 1, 2, 3).
I would like to inquire if there are any specific rules or guidelines for setting the order of bonds in LAMMPS. Currently, my workaround involves exporting a data file from MS software and manually arranging the bonds in the desired order, which doesn't seem ideal.
Any advice or guidance on establishing the correct bond order in LAMMPS would be greatly appreciated. Thank you for your time and assistance.
Best reards
Lin Jinghua

Hello researchers,
I have processed a MD simulation study of 26 amino acid amylose chain in water using gromacs. Now i want to analyze Dihedral angle for the glucose residues alpha 1-4 linkage, defined by atoms O5-C1-O4'-C4' (phi) and C1-O4'-C4'-C3' (psi).
I tried generating angular index file for dihedral angles, showing various phi angles. I also tried also check various publication but none of them have mentioned the methodology to calculate dihedral angles.
Please help me with the calculation of dihedral angles.
Thank you in anticipation.
For example, I have quercetine as a ligand and cutinase as protein. If i want to carried out md simulations with different concentration of quercetine, how to do that? I want to do md simulations by 50ng/ul of quercetine. Please help me. I'm using gromacs for simulations
I have conducted a 50 ns molecular dynamics simulation of a protein, and the trajectory contains numerous frames. I would appreciate guidance on how to create a trajectory cluster using a program compatible with Windows or online. Alternatively, if you're aware of any free and user-friendly software for performing clustering, I'd be grateful for your recommendation
Hello everyone,
I am new to the field of LAMMPS and MD simulation in general. Can anyone suggest a reliable open source software for building geometry other than LAMMPS and Material Studio? My eventual goal is to simulate tobermorite/jennite layer of structure with water layer in the middle. Currently, I am trying to build layers of graphite and water in one simulation box. I am using topotools in VMD to build the geometry but I couldn't control the number of atoms and also do not know how to merge to simulation box together.
Any help will be appreciated.
Hi,
I'm new at MD. so I want to know more about protein preparation steps.
I wanna work with gromos54a3
and
is it important to edit orientation of these residues CYS, GLN, ASN? WHY?
and
is it important to edit protonation state of these residues CYS, LYS, GLN, ASP, HIS? WHY?
We have used CaFE Software to predict the binding free energy of our protein-ligand complexes from our molecular dynamics simulations.
Fortunately the Software has run successfully however don’t know the exact meaning of the physical thermodynamic parameters calculated: a) Complex total (kcal/mol), b) Receptor (kcal/mol), c) Ligand (kcal/mol) and d) Delta (kcal/mol).
We understand that the first value “Complex” can be interpreted as the binding free of the protein-ligand complex , However, we are bit confused about the last two terms “Ligand” (c) and Delta (d) what would be the difference between them? or which is the most important parameter to determine the binding free energy of the ligand to the receptor, is that (c) or (d) parameter?
Thanks in advance and best regards,